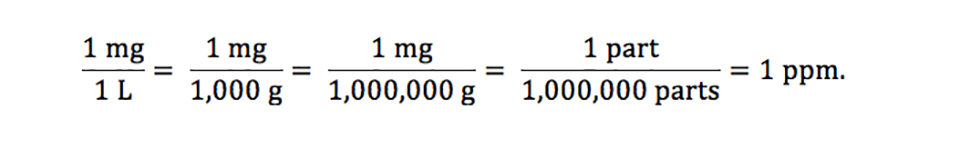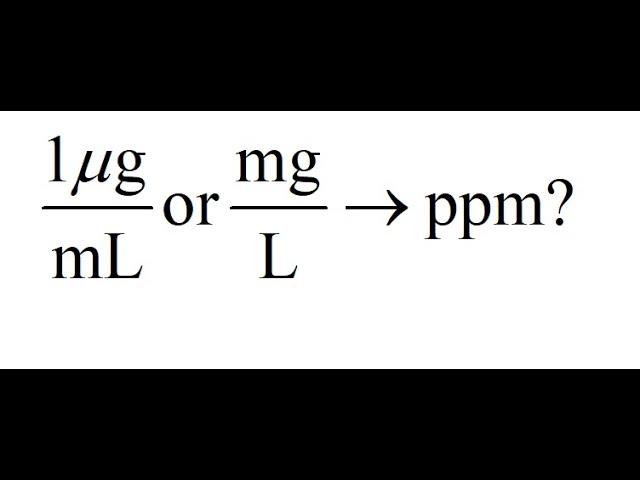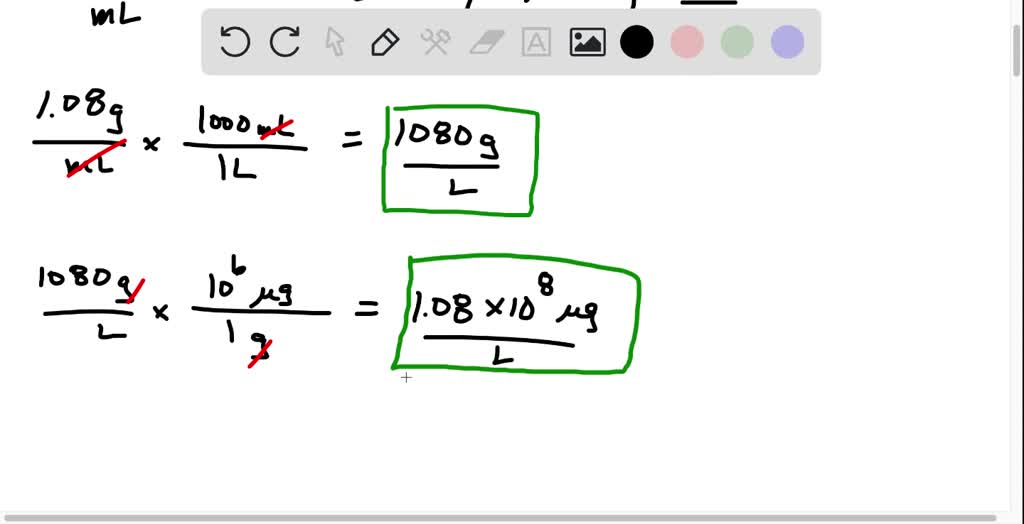
SOLVED: A dilute aqueous solution containing 1 ppm of solute has a density of 1.08 g/mL. Express the concentration of solute in g/L, μg/L, μg/mL, and mg/L.
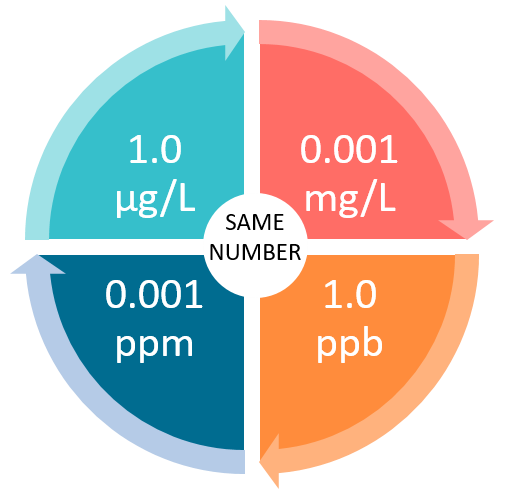
Measurement Units and Concentration Analogies 1 ppm = 1,000 ppb = 1,000,000 ppt 0.001 ppm = 1ppb = 1,000 ppt

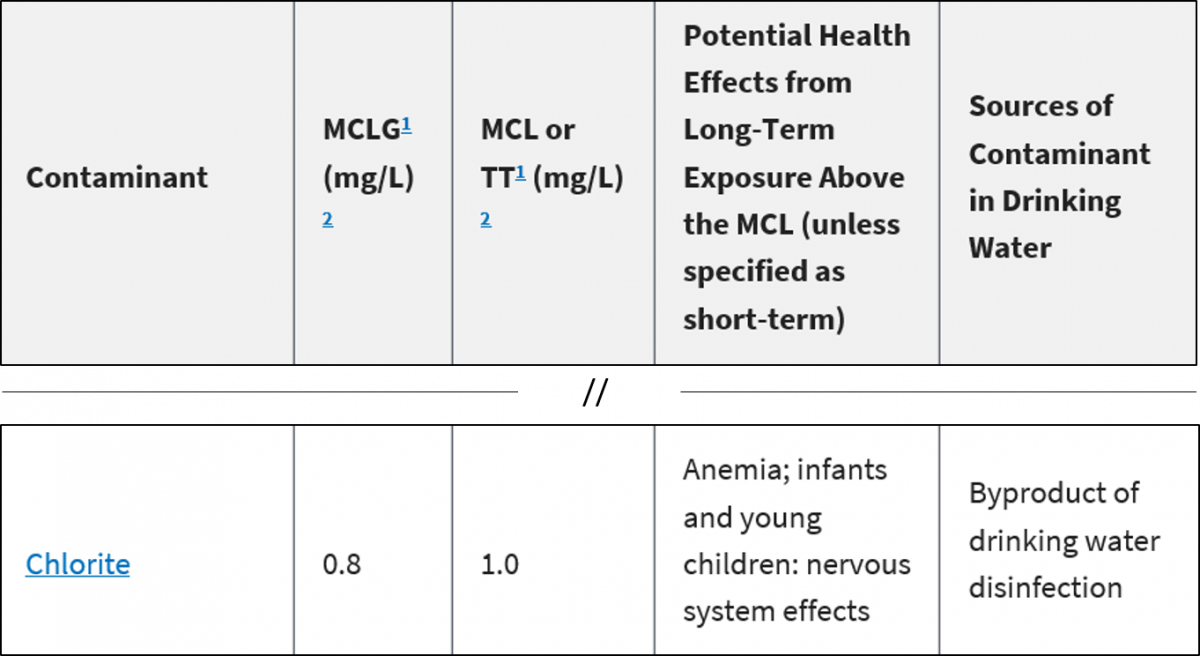
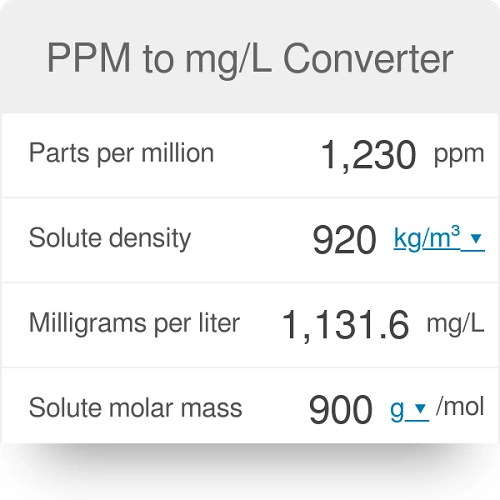
Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download
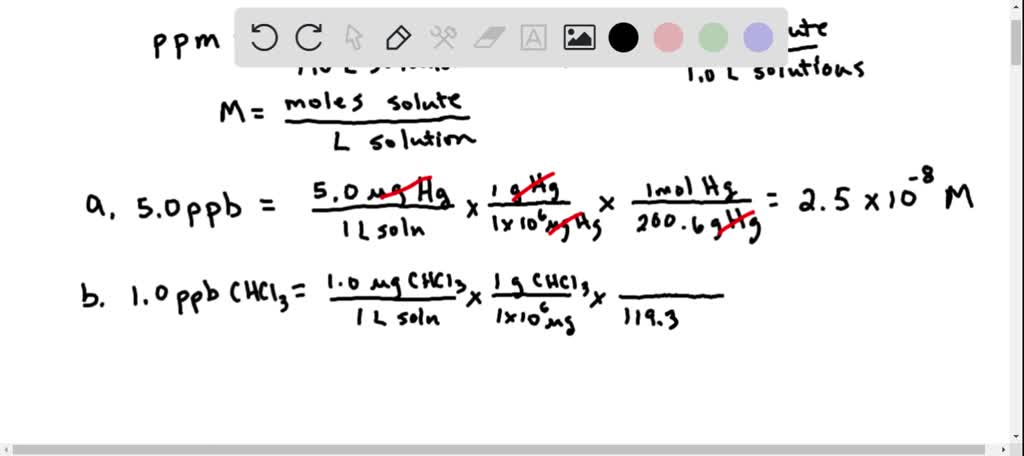
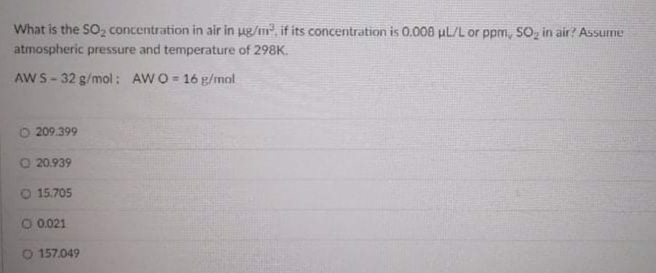
SOLVED:The units of parts per million (ppm) and parts per billion (ppb) are commonly used by environmental chemists. In general, I ppm means 1 part of solute for every 10^6 parts of

How to convert arsenic concentration ug/L (in solution) to mg/kg (in soil) after sequential extraction experiment ? | ResearchGate

How many mL of a solution of concentration 100 mg Co^2 + per mL is needed to prepare 10 mL of a solution of concentration 20 mg Co^2 + per mL ?

PPT - mg/L - milligrams per litre (10-3 g per litre) = mg per 1,000,000mg THUS = ppm PowerPoint Presentation - ID:6419103